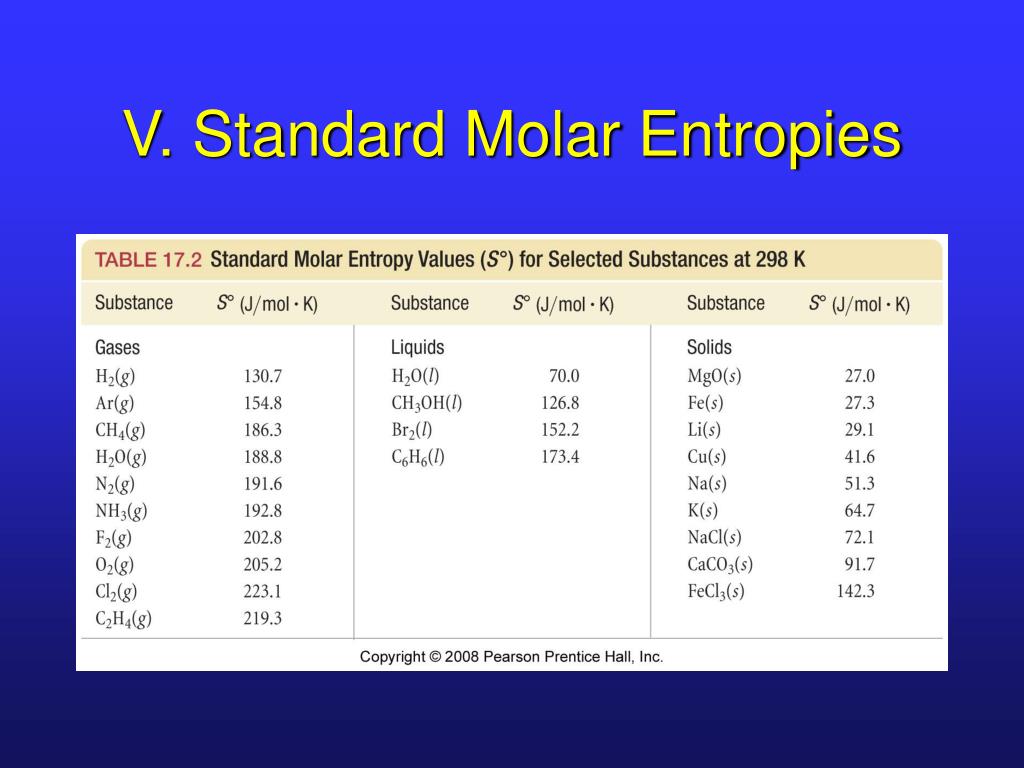
For perfectly crystalline substance entropy S = 0 at 0 KĪbsolute entropy of a pure substance : Absolute value of entropy (unlike absolute value of enthalpy) can be calculated at any temperature. 
Pure corresponds to no mixing of foriegn substances. Kinetic energy is directly proportional to Temperature, when temperature becomes zero then kinetic energy will also be zero. There are a wide variety of crystallographic defects. A perfect crystal is a crystal that contains no point, line, or planar defects. The observed molar residual entropy values for CO, RbCN, p -chloronitrobenzene, p -bromochlorobenzene, and. (9) which is equal to Rln m for the molar amounts of the above-considered substances. Third law of thermodynamics : Entropy of pure and perfectly crystalline substance at 0 K is zero. Thus, weak dipoles retain, upon freezing, the following latent entropy: S alignment (T fusion) k ln zalignment (T fusion ) N k ln mN kN ln m. 
JEE Main 2022 Question Paper Live DiscussionĪccording to third law of thermodynamics absolute value of entropy can be caculated at any temperatures. Silicon is a solid therefore, its atoms are in fixed positions, are less dispersed, and have a lower absolute molar entropy.Difference Between Selling And Marketing.TS Grewal Solutions Class 11 Accountancy.TS Grewal Solutions Class 12 Accountancy.CBSE Previous Year Question Papers Class 12.Explain why the reaction occurs only at high temperatures. (f) The reaction is thermodynamically favorable at all temperatures. (e) Calculate the value, in J/(molK), of S° for the reaction. CBSE Previous Year Question Papers Class 10 A table of absolute entropies of some substances is given below (d) Explain why the absolute molar entropy of Si(s) is less than that of H2(g).NCERT Solutions For Class 6 Social Science.NCERT Solutions for Class 7 Social Science Standard molar entropy is defined as the entropy or degree of randomness of one mole of a sample under standard state conditions.NCERT Solutions for Class 8 Social Science.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
